Synthetic Morphogenetics and Tissue Ecology Lab
Human Organoids, Synthetic Niches and Programmed Tissues

A class of synthetic living systems dubbed organoids represent 3D partial mini replicas of human organs. They provide new platform to fill the gap between mouse studies and human trials, enable precision medicine, drug discovery and human disease modeling. However, a key challenge with human organoids centers on the ability to control the process of stem cell morphogenesis and self-organization in cultures. Other challenges are limited maturation, partial vascularization, lack of common media for diverse cell types, and aberrant signatures from other organs. Additionally, tools for quantitative and universal assessment across studies are still needed.
We use synthetic biology and genetic engineering approaches to address challenges in building and controlling multicellular systems. Our approach aims to produce tissues that are functionally robust, are developed by a priori genetic design, and closely mimic their in vivo counterparts (that produced by nature). The genetically embedded circuits drive multilineage co-differentiation, direct maturation, and vascularization paving the path towards programmable organoids.
Please see References for additional information.
Case Study: Building a Human Fetal Liver
Access to fetal tissue is restricted which hamper our understanding of development. Additionally fetal liver cells are unique cell source for regenerative medicine. We have used human induced pluripotent stem cells to engineer complex multicellular systems such as human fetal liver (Fetal Liver Organoids: FeLOs). Our approach engineered a simple genetic circuit that encode GATA6 protein and triggered self-organization of human stem cells towards medoderm, endoderm and eventually vascularized FeLOs (Guye P*, Ebrahimkhani MR*, Nature Commun 2016).


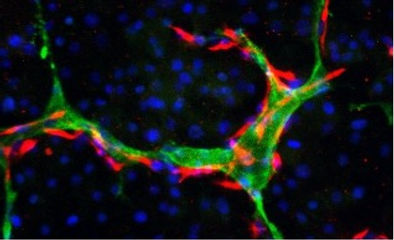
Synthetic Maturation and Vascularization of Human Liver Organoids via engineering of inter and intra-cellular gene networks

Organoids from stem cells are immature and have limited vascularization or native composition of cell types which is a major bottleneck. To address this challenge we teamed up with Kiani lab (@U Pitt) and Cahan lab (@ JHU). First we used a computational analysis and machine learning approach (CellNet: Cahan P, Cell 2014) to compare and identify potential candidate for programming of tissue towards adult liver. Next we developed cell type specific genetic circuits for multi-step activation of key pathways to produce more adult-like liver tissue. Through this approach we can promote tissue maturation to the level in range of (liver 3rd trimester of pregnancy or mature adult human liver—depending to the target genes). We showed activation of several pathways such as coagulation factors and bile acid signaling.
Genetically Guided Engineering : Computational analysis and Engineering GRNs to navigate tissue development
We showed it is possible to steer and fast-track tissue developmental stages towards native adult tissue via encoded genetic circuits in organoids.
The liver tissues generated by genetic design and engineering named as Designer Liver Organoids (DesLO), showed hepatocyte, endothelial and pericyte development. It secreted high level of human protein (e.g., albumin) in mouse blood and exhibit activation of CYP3A4 enzyme a key protein for drug metabolism. It can also model disease conditions such as tissue fibrosis. DesLOs are generate by stepwise activation of genetic circuits in human stem cells.
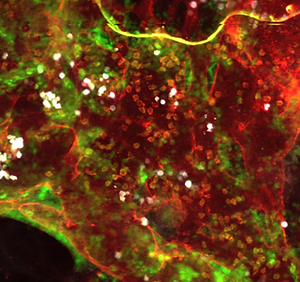

Ongoing
-
Synthetic Niches (upcoming)
-
Circuits for autonomous controlled Multi-step development of organoids (upcoming)


Selected references:
1. Synthetic Maturation of Multilineage Human Liver Organoids via Genetically Guided Engineering. bioRxiv. 2020 May 10.
Published as : Gene Regulatory Network Analysis and Engineering Directs Development and Vascularization of Multilineage Human Liver Organoids.
Velazquez JJ, et al. Cell Syst. 2021. PMID: 33290741
2. Programming Morphogenesis through Systems and Synthetic Biology. Trends in Biotechnology. 2018 Apr;36(4):415-429.
3. Genetically engineering self-organization of human pluripotent stem cells into a liver bud-like tissue using Gata6. Nat Commun. 2016 Jan 6;7:10243.
4. Approaches to in vitro tissue regeneration with application for human disease modeling and drug development. Drug Discov Today. 2014 Jun;19(6):754-62.
5. Synthetic developmental biology: build and control multicellular systems.
Curr Opin Chem Biol. 2019 Oct; 52:9-15.